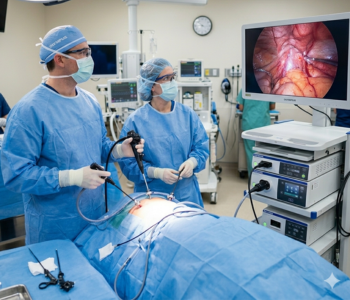
Francis Medical achieved a significant milestone in prostate cancer care by performing the first commercial Vanquish Water Vapor Ablation procedure in Austin, Texas. This procedure represents a major advancement in minimally invasive therapies for men with localized prostate cancer.
Dr. Eric Giesler of Urology Austin led the treatment at Pinnacle Surgery Center, targeting prostate tissue in a patient diagnosed with intermediate-risk, localized prostate cancer. The case reflects the patient profiles studied in the ongoing VAPOR 2 clinical trial, supporting real-world application of the technology.
Cleared by the U.S. Food and Drug Administration in November 2025, the Vanquish System offers an outpatient alternative to traditional surgery or radiation therapy. Consequently, patients can access effective treatment while avoiding extended hospital stays.
The system delivers thermal water vapor directly to the prostate using a transurethral approach guided by ultrasound and electromagnetic navigation. This approach allows precise tissue ablation and may reduce complications. Because the technology spares surrounding healthy structures, patients experience lower risks of urinary incontinence and erectile dysfunction.
The VAPOR 2 study enrolled 235 men across 26 clinical sites in the United States to evaluate the system’s safety and effectiveness in intermediate-risk, localized prostate cancer patients. Six-month biopsy results from the first 110 participants showed a 91% elimination rate of MRI-visible, biopsy-confirmed cancer after a single procedure. Importantly, no device-related serious adverse events were reported.
“Offering this innovative treatment represents a milestone in prostate cancer care,” said Dr. Giesler. He added that Vanquish delivers consistent tissue ablation while reducing side effects associated with traditional treatments. Moreover, the procedure demonstrates Urology Austin’s commitment to advanced, rigorously tested technology.
Patient feedback from VAPOR 2 was positive, with most participants reporting minimal pain and high satisfaction levels. At 12 months, 93% of patients expressed satisfaction with their outcomes. Similarly, 94% confirmed that undergoing the treatment was the right decision for their prostate cancer management.
Dr. Christopher Dixon, Francis Medical’s chief medical officer, emphasized the innovation behind Vanquish. He noted that the system could improve clinical outcomes without negatively affecting quality of life, a key concern for men facing prostate cancer treatment.
“This achievement reflects our commitment to therapies that are tough on cancer but gentle on patients,” Dixon stated. Michael Hoey founded Francis Medical in 1991 to honor his father, who died from prostate cancer. Since then, the company has focused on advancing water vapor technology to address unmet needs in urological oncology.
The Vanquish System uses thermal energy stored in sterile water vapor to ablate diseased tissue. This method offers a balance between effectiveness and preserving quality of life, which traditional treatments may compromise. Additionally, it provides a repeatable, predictable approach for prostate cancer tissue ablation.
Transitioning from clinical trials to commercial use, Vanquish procedures are designed for outpatient settings. Men can receive treatment without extended hospital stays, ensuring faster recovery. Furthermore, positive VAPOR 2 results may support future premarket approval submissions to expand indications for managing clinically localized prostate cancer.
Despite promising outcomes, FDA guidance clarifies that Vanquish is cleared as a tool for thermal ablation of targeted prostate tissue. It has not been evaluated for definitive long-term prostate cancer outcomes. Therefore, continued follow-up in clinical studies remains essential to assess treatment durability and overall disease progression.
Prostate cancer remains one of the most commonly diagnosed cancers in men worldwide. Millions face critical decisions about treatment every year. Innovations such as Vanquish offer hope for effective, minimally invasive care, reducing the burden of traditional therapies. Consequently, emerging options like water vapor ablation signal a shift toward patient-focused, personalized prostate cancer management.
With the first commercial procedure completed, Francis Medical expects broader adoption of Vanquish technology. Clinicians now have access to safe, effective alternatives for men with localized prostate cancer. Ultimately, the technology expands treatment choices and may improve patients’ quality of life across diverse care settings.